Uroplug is raising its pre-seed round to kickstart pilot batch manufacturing, develop clinical trials, and bring a breakthrough therapeutic device to market.
Over 400 million patients worldwide suffer from urinary incontinence with limited therapeutic options. Current standard of care is inadequate, archaic, or invasive, creating signficant incentive for a novel solution.
Limited existing options offer expensive, obtrusive, painful, risky, and inefficient treatments for urinary incontinence. Uroplug was designed by a team of industry-leading experts to change that.
We have a defined FDA pathway informed by pre-submission guidance, a rigorous clinical plan, and experienced medical experts, entrepreneurial innovators, and regulatory leadership on the team.
As a patient end-user device, Uroplug can reach market through a DTC and specialty physician channel strategy, eliminating the massive overhead and market quagmire of traditional medtech at a fraction of the cost.

Pre-seed capital will accelerate Uroplug through key value-inflection milestones, de-risking the program ahead of a future seed or Series A raise.
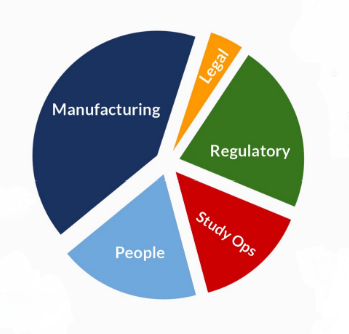 Use of Funds Pie Chart
Use of Funds Pie Chart
Close funding round to capitalize the company through first major clinical, manufacturing, and regulatory milestones.
First-in-human initial study to confirm safety and generate initial efficacy signals with the Uroplug device.
File additional provisional and non-provisional applications covering supplemental device features and use cases.
Expand investment to fund FDA engagement, additional manufacturing runs, and full-scale Feasibility study at Yale School of Medicine.
Raise follow-on capital to fund national pivotal trial, expanded product pipeline, and preparation for market entry.
Finalize mass manufacture and product commercialization in pursuit of market entry or licensure or acquisition.
Uroplug is a pre-seed stage company with awarded patents, a functional prototype, and a defined clinical development plan. We are currently finalizing our first institutional round of funding raise.
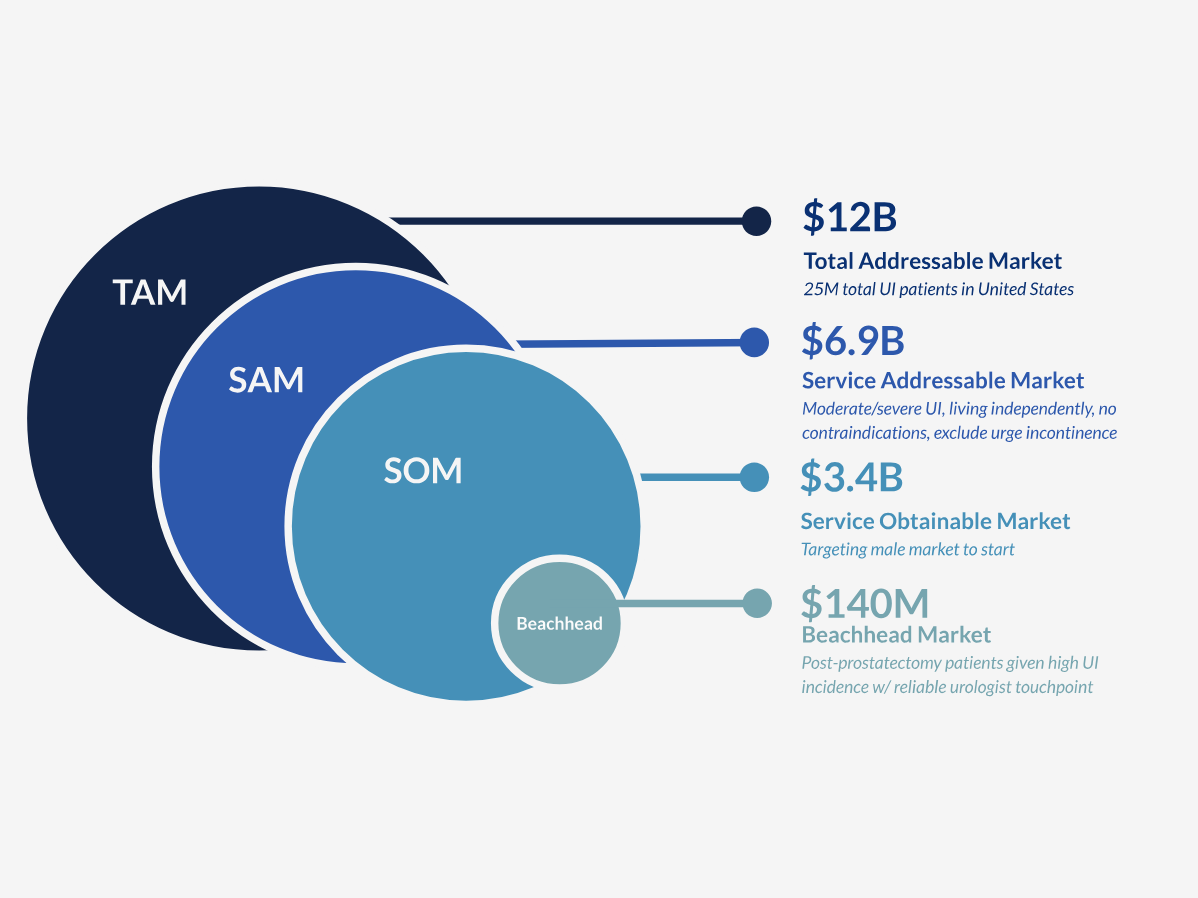
Uroplug is designed to target chronic sphinteric urinary incontinence. The condition affects 25 million patients in the US alone, with limited effective treatment options and significant unmet clinical need.
Current treatment options include deep urethral insertion devices, penile clamps and pessaries, and external urethral patches and seals. Uroplug's device offers a differentiated, non-invasive alternative with a novel mechanism of action protected by multiple patents. A detailed competitive analysis is available in our investor deck.
We are pursuing a rigorous premarket 510(k) regulatory pathway with the FDA. Our strategy has been informed by pre-submission interactions and is supported by experienced regulatory advisors.
Please reach out via the contact form below or email us directly at investors@uroplug.com. We're happy to share our deck and schedule an introductory call.
We welcome conversations with investors, strategic partners, clinicians, and anyone passionate about improving patient outcomes.
By submitting, you agree to our privacy policy. Deck shared under NDA.
Disclaimer: This website is for informational purposes only and does not constitute an offer to sell or a solicitation of an offer to buy any securities. Any offering of securities will be made only pursuant to a definitive offering document and applicable securities laws. Forward-looking statements involve risks and uncertainties. The Uroplug device is investigational and has not been cleared or approved by the FDA.